
Cutting edge of genetically
modified pigs targeting
complement activation
for xenotransplantation
Qin Sun
1
†
, Si-Yuan Song
2
†
, Jiabao Ma
3
†
, Danni Li
4
†
,
Yiping
Wang
5
, Zhengteng Yang
3
*
and Yi Wang
6
*
1
Department of Endocrinology, Sichuan Academy of Medical Sciences and Sichuan Provincial
People’s Hospital, School of Medicine, University of Electronic Science and Technology of China,
Chengdu, China,
2
Department of Neuroscience, Baylor College of Medicine, Houston, TX, United
States,
3
School of Pharmacy, Guangxi University of Chinese Medicine, Nanning, China,
4
Department
of Pharmacy, Longquanyi District of Chengdu Maternity & Child Health Care Hospital,
Chengdu, China,
5
Department of Critical Care Medicine, Sichuan Academy of Medical Sciences and
Sichuan Provincial People’s Hospital, School of Medicine, University of Electronic Science and
Technology of China, Chengdu, China,
6
Clinical Immunology Translational Medicine Key Laboratory
of Sichuan Province, Center of Organ Transplantation, Sichuan Academy of Medical Science and
Sichuan Provincial People’s Hospital, Chengdu, Sichuan, China
In the quest to address the critical shortage of donor organs for transplantation,
xenotransplantation stands out as a promising solution, offering a more abundant
supply of donor organs. Yet, its widespread clinical adoption remains hindered by
significant challenges, chief among them being immunological rejection. Central
to this issue is the role of the complement system, an essential component of
innate immunity that frequently triggers acute and chronic rejection through
hyperacute immune responses. Such responses can rapidly lead to transplant
embolism, compromising the function of the transplanted organ and ultimately
causing graft failure. This review delves into three key areas of
xenotransplantation research. It begins by examining the mechanisms through
which xenotransplantation activates both the classical and alternative
complement pathways. It then assesses the current landscape of
xenotransplantation from donor pigs, with a particular emphasis on the
innovative strides made in genetically engineering pigs to evade complement
system activation. These modifications are critical in mitigating the discordance
between pig endogenous retroviruses and human immune molecules.
Additionally, the review discusses pharmacological interventions designed to
support tra nsplantation. By exploring the intricate relationship between the
complement system and xenotransplantation, this retrospec tive analysis not
only underscores the scientific and clinical importance of this field but also
sheds light on the potential pathways to overcoming one of the major barriers to
the success of xenografts. As such, the insights offered here hold significant
promise for advancing xenotransplantation from a research concept to a viable
clinical reality.
KEYWORDS
xenotransplantation, complement systems, genetically modified pigs, C3a, C3b
Frontiers in Immunology frontiersin.org01
OPEN ACCESS
EDITED BY
Lisha Mou,
Shenzhen Second People’s Hospital, China
REVIEWED BY
Jeanette Villanueva,
Victor Chang Cardiac Research Institute,
Australia
Xiaofeng Zhang,
Greenwich Hospital/Yale New Haven Health,
United States
*CORRESPONDENCE
Yi Wang
Zhengteng Yang
†
These authors have contributed equally to
this work
RECEIVED 08 February 2024
ACCEPTED 15 March 2024
PUBLISHED 04 April 2024
CITATION
Sun Q, Song S-Y, Ma J, Li D, Wang Y, Yang Z
and
Wang Y (2024) Cutting edge of
genetically modified pigs targeting
complement activation for
xenotransplantation.
Front. Immunol. 15:1383936.
doi: 10.3389/fimmu.2024.1383936
COPYRIGHT
©2024Sun,Song,Ma,Li,Wang,Yangand
Wang. This is an open-access article distributed
under the terms of the Creative Commons
Attribution License (CC BY). The use,
distribution or reproduction in other forums
is permitted, provided the original author(s)
and the copyright owner(s) are credited and
that the original publication in this journal is
cited, in accordance with accepted academic
practice. No use, distribution or reproduction
is permitted which does not comply with
these terms.
TYPE Mini Review
PUBLISHED 04 April 2024
DOI 10.3389/fimmu.2024.1383936

1 Introduction
As of 2019, China’s organ donor registration boasted close to
1.7 million volunteers, a testament to its advancements in the field
of organ transplantation. In that same year, China ranked as the
world’s second-largest provider of allogeneic transplants,
showcasing over 10,000 kidney and 5,000 liver transplants at the
4th China-Internation al Organ Do nation Conference (1). A
significant policy shift in 2015 marked the transition to voluntary
organ donations from Chinese citizens as the exclusive legal source
for transplants (2), which, despite its ethical merits, has led to an
even greater deficit in available human organs for transplantation
and hindered research due to the scarcity.
This backd rop has propelled xenotransplantation to the
forefront as a promising solution to this shortage. Research in
this domain has progressively moved toward identifying specific
donor species, with primates being an initial choice due to their
genetic closeness to humans. However, the use of baboon organs
has consistently resulted in patient fatalities (3), steering the
scientific focus toward pigs as suitable organ donors. Pigs, with
their comparable organ size to humans and favorable breeding
traits, are currently the focal point of xenotransplantation research
(4–6). The journey of xenotransplantation, illustrated in Figure 1,is
now directed toward the development of transgenic pigs, which are
being heralded as the next step in transplantation science.
The hyperacute rejection of transplants, primarily driven by the
complement syst em, has been a longstanding ch allenge. This
system’s activation leads to the production of active compounds
like C3a and C3b (7), which catalyze immune inflammation and
graft endothelial thromboembolism. The discovery of a-Gal on
graft surfaces as a trigger for complement activation has steered the
development of a-Gal knockout (a-GalKO) pigs. Chinese research
teams, such as the one led by Pandengke, have been at the helm of
creating and refining a-Gal and b-Gal knockout pigs for several
generations. A milestone was achieved in June 2020 with t he
cloning of a pig possessing triple knockouts, a significant leap
made possible by gene editing technologies targeting the
B4GalNT2 and CMAH genes (8).
The crux of this article revolves around the utilization of gene
editing to modify pig donors, aiming to mitigate the issue of
complement activation-induced hyperacute rejection post-
xenotransplantation. We discuss dual approaches to this end: the
genetic elimination of the a-Gal epitope from x enograft
endothelium and the introduction of human complement
regulatory proteins (hCRPs) into grafts via transgenesis.
Additionally, we explore the pharmaceutical avenues developed to
inhibit the complement system, a critical strategy to counter
rejection in xenotransplantation.
2 Xenograft activates the
complement system
Xenograft transplantation challenges the human immune
system, particu larly through the activation of the co mplement
system, a sophisticated network of over 50 proteins crucial for the
immune response (9). It can be activated via three primary
pathways: the classical p athway (CL), the a lternative pathway
(AP), and the lectin pathway (MBL) (10), all leading to the
potential destruction of the xenograft.
The classical pathway is initiated by the C1 complex binding to
antigen-antibody complexes, leading to the activation of C4 and C2,
and subs equently, the formation of C3 convertase (11 ). This
enzyme is pivotal in cleaving C3 into C3a and C3b, with C3b
joining with C4b2a to form C5 convertase, advancing the
complement cascade (12). In contrast, the alternative pathway,
triggered by substances like natural polysaccharides, relies on the
spontaneous hydrolysis of C3 and the formation of a fluid-phase C3
FIGURE 1
Milestones in the history of xenotransplantation. This timeline graphically represents the pivotal moments and groundbreaking achievements in the
field of xenotransplantation from the early 20th century to modern day.
Sun et al. 10.3389/fimmu.2024.1383936
Frontiers in Immunology frontiersin.org02
convertase, leading to a modest production of C3b that enhances
phagocytosis and anaphylatoxin production (13, 14). The lectin
pathway st arts with MBL binding to microorganism surface
carbohydrates, recruiting MASP-1 and MASP-2 to form C3
convertase, mirroring the classical pathway’s initial steps (15, 16).
Xenotransplantation, especially from pig donors to primate
recipients, introduces immunologi cal hurdles due to the rapid
complement-mediated response that often leads to hyperacute
rejection (HAR), characterized by graft embolism and failure (7,
17). The presence of natural antibodies in the recipient binding to
pig endothelial cell surface glycoproteins, such as a-galactosidase
(a-Gal) and N-acetylneuraminic acid hydroxylase (Neu5Gc
protein), activates the complement system, leading to clott ing,
vascular embolism, and graft failure (18, 19). Studies have shown
that pig hearts transplanted into baboons are susceptible to this
rapid rejection, with serum analysis revealing IgM-a-Gal antibodies
bound to a-Gal, triggering the complement activation pathways
(18, 19).
However, genetic engineering offers promi sing strategies to
circumvent HAR by modifying donor pigs to reduce the human
complement system’s activation effects on graft survival. Knocking
out genes encoding heterologous endothelial antigens and creating
transgenic pigs expressing hCRPs are at the fore front of these
strategies (20). In vitro studies using pancreatic islets from a-
GalKO pigs showed reduced antibody deposition and lower levels
of complement activation, suggesting a diminished role of the lectin
pathway in xenograft rejection (18, 19).
Further research into the immunological interactions between
pig tissues and primate hosts has revealed that even in the absence
of preformed natural antibodies, HAR can occur, potentially
through the alternative complement pathway (21, 22). This
indicates a complex interplay between the classical and alternative
pathways in graft rejection, where the alternative pathway may
exacerbate C3a deposition within grafts, amplifying inflammatory
and immune responses ( 23).
Complement proteins C3a and C5a, along with the membrane
attack complex formed via the classical and alternative pathways,
play critical roles in xenograft tissue lysis. These proteins not only
mediate inflammation but also activa te coagulation casca des,
contributing to the risk of thromboembolism in xenografts (24).
Studies have shown that inflammation induced by complement
activation can significantly reduce the expression of porcine
thrombomodulin, an anti-in
flammatory molecule, on v ascular
endothelial cells, highlighting the interconnectedness of
inflammation and thrombosis in xenotransplantation (25).
Addressing the challenge of HAR in xenotransplantation
requires innovative approaches to prevent complement activation.
Genetic modifications in pig donors, such as eliminating a-Gal
epitopes and introducing hCRPs, represent vital steps toward
improving graft survival and reducing complement-mediated
rejection risks. These strat egies not o nly a im to mitigat e the
immediate immunological challenges but also open new avenues
for long-term success in xenotranspl antation, poten tially
transforming it into a viable solution for organ shortages (20).
3 Genetic modifications in pigs
Pigs are optimal donors for xenotransplantation due to their
genetic, physiological, and anatomical similarities to humans,
alongside their capability for breeding in controlled environments
(26, 27). Despite these advantages, the genetic differences between
pigs and humans can lead to im munologica l discordance and
potential organ rejection. Advancements in genetic engineering
and somatic cell nuclear transfer have facilitated modifications to
the pig genome to reduce organ immunogenicity, aiming to prevent
the human immune system from rejecting pig organ transplants
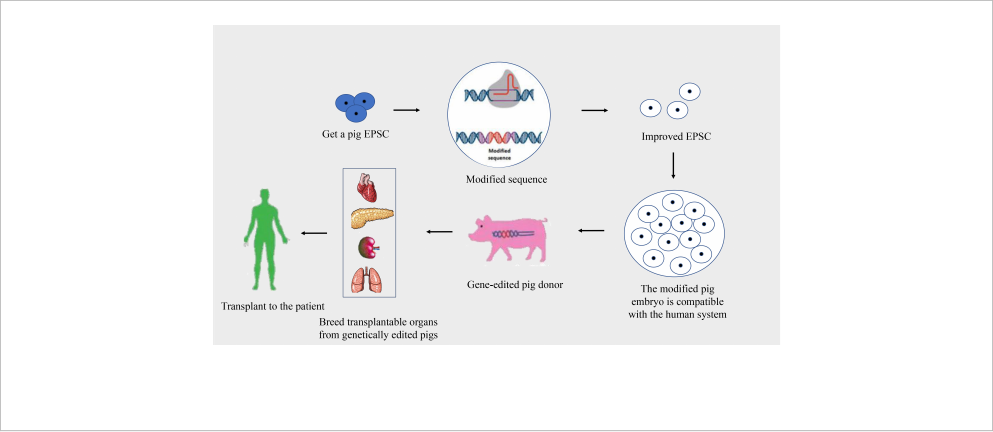
(27, 28)(Figure 2). This progress is pivotal in addressing immune
rejections, with research exploring the growth of human organs
within pigs through chimeric methods, although still
predominantly in rodent models.
The risk of viral infection, particularly from porcine
endogenous retroviruses (PERVs), represents a significant
challenge in xenotransplantation (29). Strategies to mitigate this
FIGURE 2
Process of creating gene-edited pig donors for xenotransplantation. This flowchart illustrates the stages of developing gene-edited pigs for organ
donation to human recipients.
Sun et al. 10.3389/fimmu.2024.1383936
Frontiers in Immunology frontiersin.org03
risk include breeding pigs in specifi c-pathogen-free (SPF)
environments and selecting pigs free from PERV-C to reduce the
risk of PERV-A/C-mediated transmission to humans (30).
Although endogenous ret roviruses r emain inactive within their
host species, causing no apparent disease, they could potentially
become active and infectious upon transmission to a recipient (27,
31, 32). Immune molecular incompatibility poses another obstacle,
with the immune system targeting foreign grafts, notably triggered
by pre-existing natural xenoantibodies recognizing Gal epitopes
(33–35). Genetically engineered pigs lacking alpha-1,3-Gal epitopes
represent a crucial step toward overcoming HAR and other forms of
immune rejection (27, 28).
Non-specific immune rea ctions, such as the instant blood-
mediated inflammatory reaction (IBMIR), significantly challenge
xenogeneic islet transplantation, leading to substantial graft loss
(36). Addressing these reactions involves genetic modifications of
donor animal s, anticoagul ation t herapies, and the use of anti-
inflammatory treatments to preserve graft integrity and prevent
adaptive immune activation (37).
4 Genetic modification of pigs
for xenotransplantation
The development of genetically engineered pigs marks a
significant leap forward in addressing the challenges of
xenotransplantation from pigs to primates. Through cutting-edge
genome editing techniques, scientists have been able to introduce
precise modifications into the pig genome to mitigate xenograft
rejection and diminish the risk of interspecies infection (28).
Among the most promising modifications are the disruption of
the a-Gal and the incorporation of hCRPs, which have shown
considerable promise in preclinical studies involving pig-to-non-
human primate transplants.
Recent breakthroughs in gene editing, powered by artificial
nuclease technologies, have significantly expanded the possibilities
for generating gene-edited pigs. These technologies, including zinc
finger nuclease (ZFN) (38), transcription activator-like eff ector
nuclease (TALEN) (39), and the CRISPR/Cas system (40–43),
have enabled not only simple gene knockouts and knock-ins but
also complex multi-gene editing, precision point mutations, and
conditional gene modifications. These advancements allow for gene
editing at various developmental stages of pigs, offering new
avenues for creating donor pigs with optimized genetic traits
for xenotransplantation.
The hCRPs play a crucial role in maintaining the delicate
balance between complement activation and inhibition. Proteins
such as decay-accelerating factor (hDAF), membrane cofactor
protein (hMCP), and reactive membrane cleavage inhibitor
(hCD59) prevent unregulated complement activity, which could
otherwise lead to continuous production of complement
components and exacerbate endothe lial damage in xeno grafts
(44). The expression of these hCRPs in donor pigs can
significantly reduce the risk of hyperacute rejection by limiting
the formation of th e m embrane attack complex (MAC) and
mitigating complement-mediated damage.
The application of DAF (CD55), a membrane component found
on various human cells, has been explored for its potential to protect
grafts from early rejection phases (45, 46). DAF can disrupt C3
convertases on the cell sur face, effectively downreg ulating
complement activation. Studies have demonstrated that expressing
hDAF in pig islets and other tissues can enhance protection
against human complement- mediated lysis and extend graft
survival (47, 48). Similarly, the expression of human h-transferase,
an inhibitor of the alter native complement pathway, has been
shown to provide significant protection for xenografts against
human complement atta ck, as evidenced by experiments with
transgenic pig livers transplanted into baboons (49, 50). These
genetic modifications underscore the potential of genetically
engineered pigs to overcome some of the most significant barriers
to successful xenotransplantation.
Membrane cofactor protein (MCP, CD46) plays a crucial role in
preventing the amplification loop of C3b deposition mediated by
alternative convertase. In an innovative approach, researchers
employed
a-GalKO pigs that were genetically modified to express
human CD46 across all tissues, including the heart, exhibiting
elevated levels of human CD46 expression. This genetic
modific ation n ot o nly prevented B cell infiltration but a lso
significantly reduced T cell activity in the peripheral blood of
transplants, indicating an effective suppr ession of the T cell-
mediated response to xenoantigens (51).
Human C D59 serves as a protective mechanism against
autologous cell damage by the human complement system,
specifically by inhibiting the formation of the membrane attack
complex (MAC) during the final stage of complement activation (7,
52). Utilizing embryonic germ (EG) cells, which unlike somatic cells
can proliferate indefinitely while remaining undifferentiated, Hosup
Shim (53) developed a method to create transgenic pigs capable of
expressing human CD59. These EG cells, derived from primordial
germ cells (PGC) (54), were genetically modified with a 456 bp
fragment of the hCD59 gene, encompassing the entire cod ing
region, obtained from human fibrobla st genes (55). Post-
transfection into porcine EG cells (56), these modified cells
exhibited significantly higher mitochondrial activity when
exposed to human serum containing complement, compared to
non-transgenic controls, demonstrating enhanced survival under
HAR conditions.
The development of multi-transgenic pigs offers a promising
strategy to mitigate xenograft damage more effectively. For instance,
pig cells expressing human CD59 have shown increased resistance
to lysis by human macrophages (57). Furthermore, the expression
of a1,2-fucosyltransferase (H-transferase, HT), alongside the
knockout of the a1,3-galactosyltransferase (GT) gene, presents a
viable alternative strategy. Combining gene edits to express both
hCD59 and human HT, or to achieve a-GalKO, enhances the
protective effects against human serum, thereby improving cell and
organ survival post-transplantation (58). Transgenic pigs
expressing human CD55, CD59, and H-Transferase have shown
significant reduction in complement-mediated graft destruction
(50), although these modifications alone could n ot compl etely
prevent humoral rejection, characterized by antibody deposition
and thrombotic microangiopathy. This suggests that while
Sun et al. 10.3389/fimmu.2024.1383936
Frontiers in Immunology frontiersin.org04
significant strides have been made, further research is necessary to
minimize rejection mechanisms in xenotransplantation (28).
5 Complement system target drugs
for transplantation therapy
The complement system plays a crucial role in innate immunity
and immune regulation, protecting against infections and
participating in various physiological and pathological processes
(59). Despite its protective functions, dysregulated complement
activation can contribute to detrimental effects, including
inflammation and tissue damage. A deeper understanding of the
complement system’s components and mechanisms has spurred the
development of therapeutic drugs aimed at modulating
complement activity. These drugs target v arious complement
pathways, offering potential treatments for infectious,
inflammatory, traumatic, cancerous, autoimmune, or age-related
conditions, as well as preventing transplant rejection (60).
Eculizumab, the first drug targeting the complement system,
has revolutionized the treatment landscape for diseases like
paroxysmal nocturnal hemoglobinuria (PNH), significantly
improving patient outcomes (59, 61). In the context of organ
transplantation, the complement system is implicated in several
complications, including ischemia-reperfusion injury and antibody-
mediated rejection. Therapeutic s such as C 1-1NH (Cinryze,
Berinert, Ruconest, Cetor) and Soliris are making their way into
clinical practice, showing promise but with varying efficacy levels
(62). Future research is needed to identify the most effective
complement inhibitors and devise optimal treatment strate gies.
The development programs for inhibitors targeting over a dozen
distinct complement pathways are summarized, with some already
undergoing clinical trials in both healthy volunteers and patients
(62–64). This broad spectrum of complement-targeted therapies
underscores the system’s significance across a range of medical
conditions and its potential as a therapeutic target in transplant
medicine, where controlling complement activation could mitigate
transplant rejection and improve graft survival.
6 Conclusions and perspective
The critical shortage of human organs for transplantation is a
global challenge, and xenotransplantation has emerged as a
promising approach to address this dilemma. Genetically
engineered p igs are at the forefr ont of donor option s in
xenotransplantation, offering a viable solution to the organ
shortage crisis. Advances in gene editing technologies, such as
CRISPR/Cas9, TALEN, and somatic cell nuclear transfer (SCNT),
have significantly propelled xenotransplantation research forward,
enabling precise genetic modifications in pig donors.
The complement system plays a dual role in xenotransplantation:
it is a key player in the immune response against porcine endothelial
cells following the binding of anti- porcine antibodies and
contributes to ischemia-reperfusion injury (IRI). Additionally, its
involvement in coagulation, inflammation, and the adaptive
immune response adds layers of complexity to its function in
xenograft rejection. Despite these immunobiological challenges, the
advent of genetically modified pigs, alongside an expanding array of
immunosuppressants and anti-inflammatory medications, is
progressively overcoming the hurdles faced by xenotransplantation.
Current genetic engineering efforts targeting compleme nt
regulatory mechanisms have effectively mitigated concerns
related to complement activation. However, there remains a
potential necessity for anti-complement and anti-inflammatory
interventions, especially in acute settings, to ensure the long-term
success and acceptance of xenotransplantation as a feasible solution
to the organ shortage crisis.
Author contributions
QS: Writing - original draft. QY: Writing – original draft. S-YS:
Writing – original draft. JM: Writing – original draft. DL: Writing –
original draft. YPW: Writin g – original draft. ZY: Funding
acquisition, Writing – review & editing. YW: Funding acquisition,
Writing – review & editing.
Funding
The author(s) declare that financial support was received for the
research, authorship, and/or publication of this article. This
research was supported by the National Natural Science
Foundation of China (81802504), the Sichuan Science and
Technology Bureau (2021YFH0380 and 2023YFH0010), and a
grant from Gu angxi Science and Technology Base and Talent
Project (AD23023011).
Conflict of interest
The authors declare that the research was conducted in the
absence of any commercial or financial relationships that could be
construed as a potential conflict of interest.
Publisher’s note
All claims expressed in this article are solely those of the authors and
do not necessarily represent those of their affiliated organizations, or
those of the publisher, the editors and the reviewers. Any product that
may be evaluated in this article, or claim that may be made by its
manufacturer, is not guaranteed or endorsed by the publisher.
Sun et al. 10.3389/fimmu.2024.1383936
Frontiers in Immunology frontiersin.org05
References
1. National Bureau of Statistics of China. National Economy was Generally Stable in
2019 with Main Projected Targets for Development Achieved, (Beijing, China: National
Bureau of Statistics of China) (2020). Available at: https://www.stats.gov.cn/english/
PressRelease/202001/t20200117_1723398.html.
2. Li J-H, Xu X, Wang Y-F, Xie H-Y, Ch en J-Y, Dong N-G, et al. Chinese
expert consensus on organ protection of transplantation (2022 edition). Hepatobiliary &
Pancreatic Diseases International. (2022) 21(6):516–526. doi: 10.1016/j.hbpd.2022.10.010
3. Cooper DKC. A brief history of cross-species organ transplantation. Proc
(Bayl Univ Med Cent). (2012) 25(1):49–57. doi: 10.1080/08998280.2012.11928783
4. Reardon S. First pig-to-human heart transplant: what can scientists learn? Nature.
(2022) 601:305–6. doi: 10.1038/d41586-022-00111-9
5. Montgomery RA, Stern JM, Lonze BE, Tatapudi VS, Mangiola M, Wu M, et al.
Results of two cases of pig-to-human kidney xenotransplantation. N Engl J Med. (2022)
386:1889–98. doi: 10.1056/NEJMoa2120238
6. Porrett PM, Orandi BJ, Kumar V, Houp J, Anderson D, Cozette Killian A, et al.
First clinical-grade porcine kidney xenotransplant using a human decedent model. Am
J Transplant. (2022) 22:1037–53. doi: 10.1111/ajt.16930
7. Tan LA, Yu B, Sim FC, Kishore U, Sim RB. Complement activation by
phospholipids: the interplay of factor H and C1q. Protein Cell. (2010) 001:1033–49.
doi: 10.1007/s13238-010-0125-8
8. Zhang Y, Pan D, Sun X, Sun G, Wang X, Liu X, et al. Production of porcine cloned
transg enic embryos expressing green fluorescent protein by somatic cell nuclear
transfer. Sci China Ser C. (2006) 49):1–8. doi: 10.1007/s11427-005-0071-5
9. Liu C-CM, Susan M, Kao AH, Navratil JS, Ahearn JM. Cell-bound complement
biomarkers for systemic lupus erythematosus: from benchtop to bedside. Rheum Dis
Clin North Am. (2010) 36:161–72. doi: 10.1016/j.rdc.2009.12.003
10. Roos A, Daha M. Antibody-mediated activation of the classical complement
pathway in xenograft rejection. Transplant Immunol. (2002) 9:257–70. doi: 10.1016/
S0966-3274(02)00042-4
11. Wallis R, Mitchell DA, Schmid R, Schwaeble WJ, Keeble AH. Paths reunited:
initiation of the classical and lectin pathwa ys of complement activ ation.
Immunobiology. (2010) 1:1–11. doi: 10.1016/j.imbio.2009.08.006
12. Ballow M. C1-Bypass complement-activation pathway in patiente with chronic
urticaria and angiosoelig. Lancet. (1975) 306:248–50. doi: 10.1016/S0140-6736(75)90963-0
13. Gtze O, Müller-Eberhard HJ. The alternative pathway of complement activation.
Adv Immunol. (1976) 24:1–35. doi: 10.1016/s0065-2776(08)60328-4
14. Merle NS, Elizabeth CS, Veronique FB, Roumenina LT. Complement system
part I a. Front Immunol. (2015) 6:1–30. doi: 10.3389/fimmu.2015.00257
15. Cooper DKC, Sachs DH, Colvin RB, Shimizu A, Hisashi Y, Yamada K, et al.
Rejection of cardiac xenografts transplanted from ar,3-3omsplantedFDSLHSCRHJ
genesplantedF (GalT -alT pig s to baboons. Am J Trans plant. (2008) 8:2516–26.
doi: 10.1111/j.1600-6143.2008.02444.x
16. Kaplon RJ, Platt JL, Kwiatkowski PA, Edwards NM, Xu HE, Shah AS, et al.
Absence of hyperacute rejection in pig-to-primate orthotopic pulmonary xenografts.
Transplantation. (1995) 59:410. doi: 10.1097/00007890-199502150-00017
17. Chen RH, Kadner A, Mitchell RN, Adams DH. Mechanism of delayed rejection
in transgenic pig-to-primate cardiac xenotransplantation. J Surg Res. (2000) 90:119–25.
doi: 10.1006/jsre.2000.5864
18. Thompson P, Badell I, Lowe M, Cano J, Song M, Leopardi F, et al. Islet
xenotransplantation using gal-deficient neonatal donors improves engraftment and
function. other. (2011) 11:1–20. doi: 10.1111/j.1600-6143.2011.03720.x
19. Broom C, Uknis ME. Methods of treating antibody-mediated rejection in organ
transplant patients with C1-esterase inhibitor. (Australia: Patent) (2018).
20. Butler JR, Ladowski JM, Martens GR, Tector M, Tector AJ. Recent advances in
genome editing and creation of genetically modified pigs. Int J Surgery. (2015) 23:217–22.
doi: 10.1016/j.ijsu.2015.07.684
21.SuckfüllM,MüdsamM,PieskeO,EndersG,BabicR,HammerC.
Immunohistological studies of complement activation after xenogeneic perfusion of
a working heart model. Transplant Int. (1994) 7:324–8. doi: 10.1111/j.1432-
2277.1994.tb01241.x
22. Forty J, Hasan R, Cary N, White DJ, Wallwork J. Hyperacute rejection of rabbit
hearts by human blood is mediated by the alternative pathway of complement.
Transplant Proc. (1992) 24:488.
23. Platts-Mills TAE, Ishizaka K. Activation of the alternative pathway of human
complement by rabbit cells. J Immunol . (1974) 113:348– 58. doi: 10.4049/
jimmunol.113.1.348
24. Li Y, Gong P, Kong C, Tian X. Bufalin engages in RIP1-dependent and ROS-
dependent programmed necroptosis in breast cancer cells by targeting the RIP1/RIP3/
PGAM5 pathway. Anti Cancer Drugs. (2019) 30:e0770. doi: 10.1097/
CAD.0000000000000770
25. Ochando J, Ordikhani F, Boros P, Jordan S. The innate immune response to
allotransplants: mechanisms and therapeutic potentials. Cell Mol Immunol. (2019)
16):350–6. doi: 10.1038/s41423-019-0216-2
26. Heng Z, Kaixiang X, Ninglin F, Hongye Z, Hongjiang W. Construction and
current status of gene-edited xenotransplantation pigs. Electronic J Pract Organ
Transplantation. (2018) 6:412–8.
27. Hryhorowicz M, Zeyland J, Słomski R, Lipin
́
ski D. Genetically modified pigs as
organ donors fo r xen ot ran sp lan ta tio n. Mol Biotechnol. (2017) 59(9-10):435–44.
doi: 10.1007/s12033-017-0024-9
28. Klymiuk N, Aigner B, Brem G, Wolf E. Genetic modification of pigs as organ
donors for xenotransplantation. Mol Reprod Dev. (2010) 77:209–21. doi: 10.1002/
mrd.21127
29. Specke V, Rubant S, Denner J. Productive infection of human primary cells and
cell lines with porcine endogenous retroviruses. Vi rology. (2001) 2 85:177–80.
doi: 10.1006/viro.2001.0934
30. Patience C, Switzer WM, Takeuchi Y, Griffiths DJ, Weiss RA. Multiple groups of
novel retroviral genomes in pigs and related species. JVirol. (2001) 75:2771–5.
doi: 10.1128/JVI.75.6.2771-2775.2001
31. Denner J. Porcine Endogenous Retroviruses and Xenotransplantation. Viruses.
(2021) 13(11):2156. doi: 10.3390/v13112156
32. Denner J. Recombinant porcine endogenous retroviruses (PERVviruse a new
risk for xenotransplantation? Xenotransplantation. (2010) 17:120–0. doi: 10.1111/
j.1399-3089.2010.00573_21.x
33. Ibrahim Z, Busch J, Awwad M, Wagner R, Wells K, Cooper DKC. Selected
physiologic compatibilities and incompatibilities between human and porcine organ
systems. Xenotransplantation. (2010) 13:488– 99. doi: 10.1111/j.1399-
3089.2006.00346.x
34. Guoling L, Zhiqian X, Huaqiang Y, Zhenfang W. Research progress of transgenic
and gene-edited pigs. J South China Agric Univ . (2019) 40(5):91– 101.
35. Tanihara F, Hirata M, Otoi T. Current status of the application of gene editing in
pigs. J Reprod Dev. (2021) 67(3):177–87. doi: 10.1262/jrd.2021-025
36. Matsumoto S, Tomiya M, Sawamoto O. Current status and future of clinical islet
xenotransplantation. J Diabetes. (2016) 8(4):483–93. doi: 10.1111/1753-0407.12395
37. Zhengzhao L, Tian H, Zhiming C, Lisha M. Research progress of porcine islet
xenotransplantation. Organ transplant. (2017) 008:246–50.
38. Miller JC, Holmes MC, Wang J, Guschin DY, Lee YL, Rupniewski I, et al. An
improved zinc-finger nuclease architecture for highly specific genome editing. Nat
Biotechnol. (2007) 25:778–85. doi: 10.1038/nbt1319
39. Schmid-Burgk JL, Schmidt T, Kaiser V, Höning K, Hornung V. A ligation-
independent c loning technique for high-throughput assembly of transcription
activator-like effector genes. Nat Biotechnol
. (2013) 31(1):76–81. doi: 10.1038/nbt.2460
40. Mali P, Yang L, Esvelt KM, Aach J, Guell M, Dicarlo JE, et al. RNA-guided
human genome engineering via cas9. Science. (2013) 339 :823. doi : 10.1126/
science.1232033
41. Hsu PD, Lander ES, Zhang F. Development and applications of CRISPR-cas9 for
genome engineering. (2014) 157(6):1262–78. doi: 10.1016/j.cell.2014.05.010
42. Gaj T, Gersbach CA, Barbas CF. ZFN, TALEN, and CRISPR/Cas-based methods
for genome engineering. Trends Biotechnol. (2013) 31(7):397–40 5. doi: 10.1016/
j.tibtech.2013.04.004
43. Yaoqiang H, Guoling L, Huaqiang Y, Zhenfang W. Application of gene-edited
pigs in biomedical research. Genetic. (2018) v.40:30–44.
44. Roumenina LT, Zuber J, Fre
́
meaux-Bacchi V. Physiological and therapeutic
complement regulators in kidney transplantation. Curr Opin Organ Transplant. (2013)
18:421–9. doi: 10.1097/MOT.0b013e32836370ce
45. Kinoshita T. Distribution of decay-accelerating factor in the peripheral blood of
normal individuals and patients with paroxysmal nocturnal hemoglobinuria. J Exp
Med. (1985) 162:75–92. doi: 10.1084/jem.162.1.75
46. Asch AS. Decay-accelerating factor is present on cultured human umbilical vein
endothelial cells. J Exp Med. (1986) 163:221–6. doi: 10.1084/jem.163.1.221
47. Yamamoto T, Iwase H, King TW, Hara H, Cooper DKC. Skin
xenotransplantation: Hist orical review and clin ical potential. Burns. (2018) 44
(7):1738–1749. doi: 10.1016/j.burns.2018.02.029
48. Schmidt P, Goto M, Mauff BL, Anegon I, Korsgren O. Adenovirus-mediated
expression of human CD55 or CD59 protects adult porcine islets from complement-
mediated cell lysis by human serum. Transplantation. (2003) 75:697–702. doi: 10.1097/
01.TP.0000053249.39753.D6
49. Young-Hee J, Chi-Hun P, Gun-Hyuk J, Yeun-Ik J, In-Sung H, Yeon-Woo J, et al.
Production of multiple transgenic yucatan miniature pigs expressing human
complement regulatory factors, human CD55, CD59, and H-transferase genes. PloS
One. (2013) 8:e63241. doi: 10.1371/journal.pone.0063241
50. Ramı
́
rez P, Montoya MJ, Rı
́
os A, Palenciano CG, Majado M, Cha
́
vez R, et al.
Prevention of hyperacute rejection in a model of orthotopic liver xenotransplantation
from pig to baboon using polytransgenic pig livers (CD55, CD59, and H-transferase).
Transplant Proc. (2005) 37:4103–6. doi: 10.1016/j.transproceed.2005.09.186
51. Mohiuddin MM, Corcoran PC, Singh AK, Azimzadeh A, Hoyt RF Jr., Thomas
ML, et al. B-cell depletion extends the survival of GTKO.hCD46Tgpig heart xenografts
Sun et al. 10.3389/fimmu.2024.1383936
Frontiers in Immunology frontiersin.org06
in baboons for up to 8 months. Am J Transplantation. (2012) 12:763–71. doi: 10.1111/
j.1600-6143.2011.03846.x
52. Kimberley FC, Sivasankar B, Morgan BP. Alternative roles for CD59. Mol
Immunol. (2007) 44:73–81. doi: 10.1016/j.molimm.2006.06.019
53. Ahn KS, Ji YW, Park JK, Sorrell AM, Heo SY, Kang JH, et al. Production of
human CD59-transgenic pigs by embryonic germ cell nuclear transfer. Biochem
Biophys Res Commun. (2010) 400:667–72. doi: 10.1016/j.bbrc.2010.08.125
54. Uszewski KM. Control of the complement system. Adv Immunol. (1996) 61:201–
83. doi: 10.1016/S0065-2776(08)60868-8
55. Ji YW, Ahn KS, Sorrell AM, Shin S, Shim H. Cytolytic assessment of hyperacute
rejection and production of nuclear transfer embryos using hCD46-transgenic porcine
embryonic germ cells. Zygote. (2009) 17:101–8. doi: 10.1017/S096719940800511X
56. Lee JH, Lee HJ, Nahm KM, Jeon HY, Hwang WS, Paik NW, et al. Effects of
combined expression of human complement regulatory proteins and H-transferase on
the inhibition of complement-mediated cytolysis in porcine embryonic fibroblasts.
Transplant Proc. (2006) 38:1618–21. doi: 10.1016/j.transproceed.2006.02.129
57. Shim H. Isolation of pluripotent stem cells from cultured porcine primordial
germ cells. Biol Reproduction. (1997) 47:1089–95. doi: 10.1095/biolreprod57.5.1089
58. Chen CG, Salvaris EJ, Romanella M, Aminian A, Pearse MJ. Transgenic
expression of human alpha1,2-fucosyl transferase (H-transferase) prolongs mouse
heart survival in an ex vivo model of xenograft rejection. Transplantation. (1998)
65:832. doi: 10.1097/00007890-199803270-00011
59. Rother RP, R ollins SA, Mojcik C F, Brods ky RA, Bell L. Disc overy and
development of the complement inhibitor eculizumab for the treatment of
paroxysmal nocturnal hemoglobinuria. Nat Biotechnol. (2007) 25:1256. doi: 10.1038/
nbt1344
60. Melis JPM, Strumane K, Ruuls SR, Beurskens FJ, Schuurman J, Parren PWHI.
Complement in therapy and disease: Regulating the complement system with antibody-
based therapeutics. Mol Immunol. (2015) 67:117– 30. doi: 10.1016/
j.molimm.2015.01.028
61. Woodruff TM, Nandakumar KS, Tedesco F. Inhibiting the C5-C5a receptor axis.
Mol Immunol. (2011) 48:1631–42. doi: 10.1016/j.molimm.2011.04.014
62. Ricklin D, Lambris JD. New milestones ahead in complement-targeted therapy.
Semin Immunol. (2016) 28(3):208–22. doi: 10.1016/j.smim.2016.06.001
63. Morgan BP, Harris CL. Complement, a target for therapy in inflammatory and
degenerative diseases. Nat Rev Drug Discovery. (2015) 14(12):857–77. doi: 10.1038/
nrd4657
64. Risitano AM, Marotta S. Therapeutic complement inhibition in complement-
mediated hemolytic anemias: Past, present and future. Semin Immunol. (2016) 28
(3):223–40. doi: 10.1016/j.smim.2016.05.001
Sun et al. 10.3389/fimmu.2024.1383936
Frontiers in Immunology frontiersin.org07
